 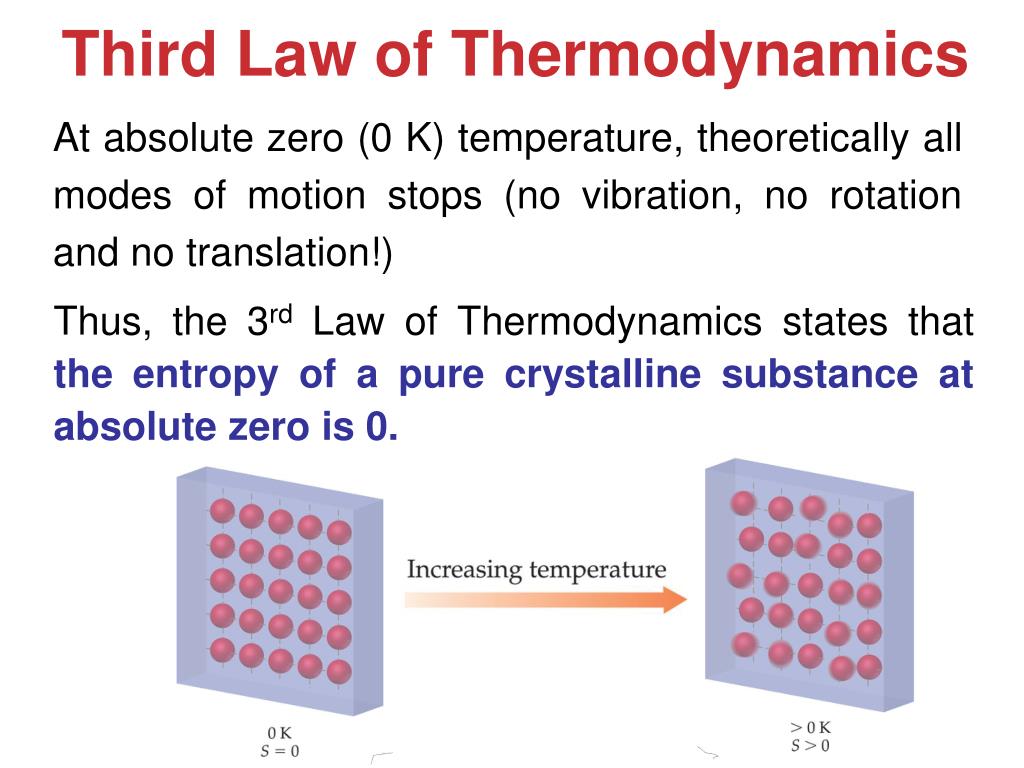 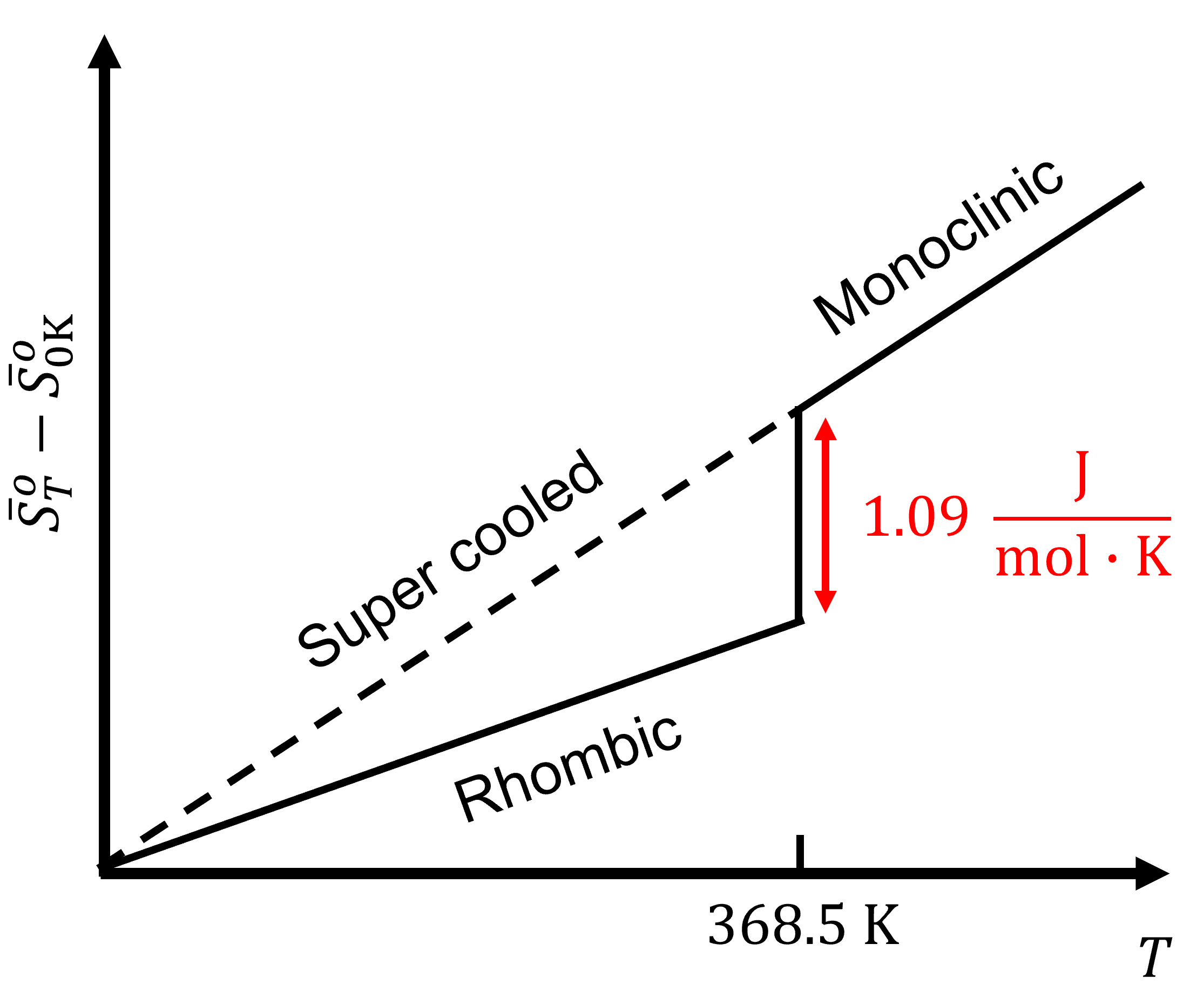
Miller, Computational Methods of Neutron Transport, American Nuclear Society, 1993, ISBN: 2-4. Hetrick, Dynamics of Nuclear Reactors, American Nuclear Society, 1993, ISBN: 3-2. To obtain the absolute value, we need the third law of thermodynamics, which states that S 0 at absolute zero for perfect crystals. Neuhold, Introductory Nuclear Reactor Dynamics, American Nuclear Society, 1985, ISBN: 9-4. With this we can only obtain the difference of entropy by integrating the above formula. It seems like the 3rd law is really just saying that, as the temperature of a closed system goes to absolute zero, the entropy and energy must both approach minimum values. Bezella, Introductory Nuclear Reactor Statics, American Nuclear Society, Revised edition (1989), 1989, ISBN: 3-2. At absolute zero (zero kelvins) the system must be in a state with the minimum possible energy. Specifically, the entropy of a pure crystalline substance. The entropy of a system at absolute zero is typically zero, and in all cases is determined only by the number of different ground states it has. First, a perfect crystal means that there are. The third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature approaches absolute zero. Department of Energy, Nuclear Physics and Reactor Theory. DOE Fundamentals Handbook, Volume 1 and 2. January 1993. The Third Law of Thermodynamics states that a perfect crystal at zero Kelvin (absolute zero) has zero entropy. Robert Reed Burn, Introduction to Nuclear Reactor Operation, 1988.Physics of Nuclear Kinetics. Addison-Wesley Pub. Nuclear and Particle Physics. Clarendon Press 1 edition, 1991, ISBN: 978-0198520467 Nuclear Reactor Engineering: Reactor Systems Engineering, Springer 4th edition, 1994, ISBN: 978-0412985317 Stacey, Nuclear Reactor Physics, John Wiley & Sons, 2001, ISBN: 0- 471-39127-1. Baratta, Introduction to Nuclear Engineering, 3d ed., Prentice-Hall, 2001, ISBN: 8-1. Lamarsh, Introduction to Nuclear Reactor Theory, 2nd ed., Addison-Wesley, Reading, MA (1983). Absolute zero is denoted as 0 K on the Kelvin scale, −273.15 ☌ on the Celsius scale, and −459.67 ☏ on the Fahrenheit scale. 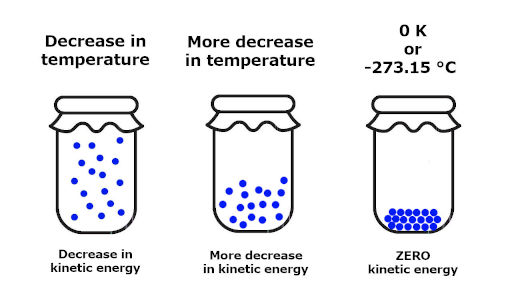
Classically, this would be a state of motionlessness, but quantum uncertainty dictates that the particles still possess a finite zero-point energy. This is a state at which the enthalpy and entropy of a cooled ideal gas reaches its minimum value, taken as 0. This allows us to define a zero point for the thermal energy of a body.Ībsolute zero is the coldest theoretical temperature, at which the thermal motion of atoms and molecules reaches its minimum. The entropy of a system approaches a constant value as the temperature approaches absolute zero.īased on empirical evidence, this law states that the entropy of a pure crystalline substance is zero at the absolute zero of temperature, 0 K and that it is impossible by means of any process, no matter how idealized, to reduce the temperature of a system to absolute zero in a finite number of steps. Absolute zero is the lowest theoretical temperature, which scientists have defined as minus 459.67 degrees Fahrenheit (minus 273.15 degrees Celsius). According to third law of thermodynamics: 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
